Tackling Biofilms: A Guide for Infectious Disease Specialists
Published on January 19, 2026 by Admin

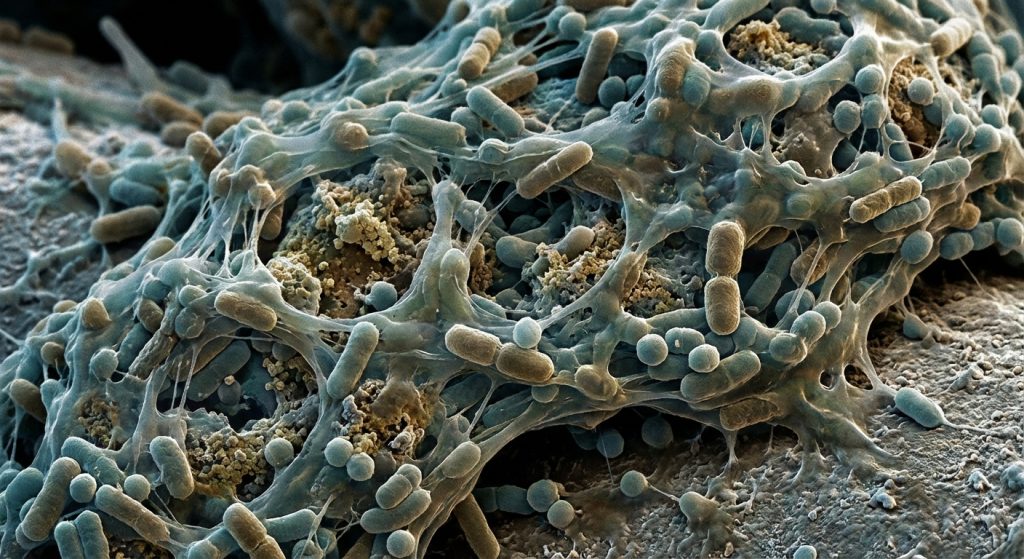
Biofilms pose a significant challenge in healthcare. They are communities of microorganisms. These microbes stick to surfaces. They also embed themselves in a protective matrix. This matrix is often self-produced. Consequently, biofilms are notoriously difficult to eradicate. They are also resistant to many standard treatments. This article explores biofilm formation. It also details current and emerging eradication strategies. Understanding these aspects is crucial for infectious disease specialists.
What Are Biofilms?
Biofilms are more than just a collection of microbes. They are structured, cooperative communities. Microorganisms, such as bacteria, fungi, and even protozoa, can form them. They typically adhere to living or non-living surfaces. Examples include medical devices and tissues. The microbes within a biofilm communicate. They also coordinate their activities. This coordination is key to their survival. It allows them to function collectively. Therefore, a single bacterium in a biofilm is very different from one floating freely.
The Biofilm Matrix
The extracellular polymeric substance (EPS) matrix is vital. It encases the microbial community. This matrix is a complex mixture. It contains polysaccharides, proteins, nucleic acids, and lipids. The EPS matrix serves many functions. For instance, it helps anchor the biofilm. It also protects the microbes. This protection is from physical forces and environmental stresses. Furthermore, it acts as a barrier. This barrier prevents antibiotics and immune cells from reaching the microbes. As a result, it contributes significantly to antimicrobial resistance.
Why Are Biofilms So Problematic?
Biofilms are a major cause of persistent infections. They are also linked to device-associated infections. Traditional antibiotics often fail against them. This failure is due to several factors. Firstly, the EPS matrix hinders drug penetration. Secondly, microbes within a biofilm can exist in a slower metabolic state. This makes them less susceptible to drugs targeting active growth. Thirdly, specific genes can be activated within the biofilm. These genes confer resistance. Thus, infections caused by biofilms are often chronic. They can also be difficult to treat effectively.
Common Biofilm-Related Infections
- Catheter-associated urinary tract infections (CAUTIs).
- Central venous catheter infections.
- Ventilator-associated pneumonia (VAP).
- Periodontal disease.
- Chronic wound infections.
- Prosthetic joint infections.
- Endocarditis.
These infections can lead to severe complications. They can also increase morbidity and mortality. Therefore, effective biofilm eradication is paramount.
Strategies for Biofilm Eradication
Combating biofilms requires a multifaceted approach. No single strategy is universally effective. However, combining different methods can improve outcomes. We can categorize these strategies broadly.
1. Antimicrobial Therapies
While standard antibiotics are often insufficient, certain antimicrobial strategies show promise. These include higher doses and longer treatment durations. However, this approach carries risks. It can also lead to increased side effects and resistance. Therefore, novel antimicrobial agents are being developed.
Antibiotics with Enhanced Penetration
Some antibiotics are better at penetrating the EPS matrix. Examples include rifampicin and daptomycin. These are often used in combination therapy. They target specific biofilm components or microbes. For example, combining a biofilm-disrupting agent with a standard antibiotic can be effective. This dual action is key.
Antibiotics Targeting Quorum Sensing
Quorum sensing is a cell-to-cell communication system. Microbes use it to coordinate behavior. This includes biofilm formation. Inhibiting quorum sensing can disrupt biofilm development. It can also make microbes more susceptible to antibiotics. This is an emerging area of research.
2. Physical and Mechanical Methods
These methods aim to physically remove or disrupt the biofilm. They are often used in conjunction with antimicrobial therapies.
Device Modification and Replacement
For device-associated infections, removing or replacing the infected device is often necessary. This is a straightforward but effective method. For example, removing an infected catheter is crucial. It prevents further microbial proliferation. This is especially important in preventing conditions like CAUTIs.
Mechanical Disruption
Techniques like sonication can physically break down biofilms. Ultrasound waves can dislodge microbes from surfaces. This can enhance antibiotic penetration. It is particularly useful for hard-to-treat infections. It can also be applied to implants.
3. Novel and Emerging Strategies
The fight against biofilms is constantly evolving. Researchers are exploring innovative approaches.
Phage Therapy
Bacteriophages are viruses that infect bacteria. They are highly specific. Phage therapy uses these viruses to target and kill bacteria. They can be particularly effective against antibiotic-resistant strains. Phages can also penetrate biofilms. They can even replicate within them. This offers a powerful new weapon. Indeed, phage therapy is revolutionizing care in many areas.
Enzymatic Disruption
Certain enzymes can degrade the EPS matrix. For example, DNases break down DNA in the matrix. Proteases break down proteins. Using these enzymes can weaken the biofilm structure. This then allows antibiotics to work more effectively. It is a promising adjunct therapy.
Antimicrobial Peptides (AMPs)
AMPs are natural molecules. They are part of the innate immune system. They can kill microbes directly. They can also disrupt biofilms. Some AMPs can even prevent biofilm formation. Their broad-spectrum activity and low resistance development make them attractive.
Quorum Quenching
This is related to quorum sensing inhibition. Quorum quenching actively degrades the signaling molecules. This prevents microbes from communicating. Consequently, it disrupts biofilm formation and maturation. It offers a non-lethal way to manage biofilms.
Probiotics and Prebiotics
Beneficial bacteria (probiotics) can compete with pathogens. They can also produce antimicrobial substances. Prebiotics provide food for these beneficial bacteria. This approach aims to restore a healthy microbial balance. It can prevent pathogens from establishing biofilms. This is particularly relevant for gut and wound infections. Improving gut health is crucial for overall immunity. You can learn more about how your gut bacteria dictate physical and mental immunity.
Antivirulence Strategies
Instead of killing microbes directly, these strategies target virulence factors. These are molecules that make microbes harmful. By disarming them, we reduce their ability to cause disease. This can also reduce the selective pressure for resistance. It is a gentler approach.
Nanotechnology-Based Approaches
Nanoparticles can be engineered. They can deliver antimicrobial agents directly to biofilms. They can also possess inherent antimicrobial properties. Their small size allows for better penetration. Furthermore, they can be designed to target specific biofilm components. This offers precision in treatment.
Prevention is Key
While eradication strategies are vital, prevention is always better. Several measures can reduce the risk of biofilm formation.
- Strict adherence to infection control protocols.
- Minimizing the use of indwelling medical devices.
- Careful selection and maintenance of medical devices.
- Regular cleaning and disinfection of surfaces.
- Promoting good oral hygiene.
- Early detection and management of wounds.
These preventive measures are fundamental. They reduce the overall burden of biofilm-related infections.
Challenges and Future Directions
Despite advances, significant challenges remain. Biofilms are diverse. They adapt quickly. Therefore, developing universally effective treatments is difficult. Furthermore, diagnosing biofilm infections can be challenging. They often present with non-specific symptoms. This can delay appropriate treatment.
The future of biofilm eradication lies in personalized medicine. Tailoring treatments to specific biofilm compositions and host factors is crucial. Combination therapies will likely become more sophisticated. Integrating various strategies—antimicrobials, physical methods, and novel agents—will be essential. Understanding the complex genetic and molecular mechanisms of biofilm formation is also key. This knowledge will pave the way for more targeted interventions. The field of antimicrobial resistance is a constant challenge. Therefore, exploring alternatives to traditional antibiotics is vital. Technologies like CRISPR are also showing promise in targeting microbial resistance mechanisms. You can explore how CRISPR tools target superbugs.
Frequently Asked Questions (FAQ)
What is the most common site for biofilm formation in hospitals?
The most common sites include indwelling medical devices like urinary catheters, central venous catheters, and endotracheal tubes. Biofilms also frequently form on heart valves and prosthetic joints.
Can biofilms be completely eliminated once formed?
Complete elimination can be very difficult, especially in chronic infections or on implanted devices. The goal is often to reduce the biofilm burden significantly and suppress the infection, sometimes requiring long-term treatment or device removal.
Are biofilms a problem only in humans?
No, biofilms are a widespread phenomenon. They affect various environments, including industrial settings (e.g., pipelines), natural water systems, and even dental plaque on animals. They are a significant concern in veterinary medicine as well.
How does quorum sensing contribute to biofilm resistance?
Quorum sensing allows bacteria to coordinate gene expression. This includes genes that enhance biofilm structure and those that confer resistance to antibiotics or host defenses. By sensing population density, they can collectively activate protective mechanisms.
What is the role of the immune system against biofilms?
The immune system attempts to combat biofilms, but its effectiveness is often limited. Immune cells like neutrophils can struggle to penetrate the biofilm matrix. Furthermore, microbes within biofilms can evade immune detection or even modulate immune responses.
Conclusion
Biofilms represent a formidable obstacle in infectious disease management. Their inherent resistance mechanisms necessitate a departure from conventional treatment paradigms. By understanding the intricacies of biofilm formation and employing a diverse array of eradication strategies—from enhanced antimicrobial therapies to cutting-edge approaches like phage therapy and nanotechnology—specialists can improve patient outcomes. Moreover, prioritizing prevention through robust infection control measures remains paramount. The ongoing research and development in this field offer hope for more effective solutions in the future. Ultimately, a proactive and integrated approach is essential to combat the persistent threat of biofilms.

